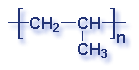Chemistry of Plastics
A Short Chemistry Lesson
The idea of plastic came from naturally occurring substances known as polymers. Polymers can be found existing in nature in the form of tar, shellac, tortoise shells, horns, tree saps such as amber and latex.
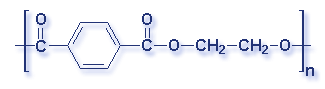
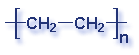
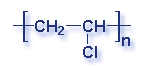
What is a polymer? A polymer is a substance made of many repeating chemical units or molecules. A simpler way to think of polymers is as a paper clip chain. The paper clips are all made out of the same thing, and as they are linked together it illustrates the concept of polymer's chemical unit repeating itself. Depending on the substances used, the polymer created may vary. Most polymers contain carbon and hydrogen, though they can also have oxygen, chlorine, fluorine, nitrogen, silicon, phosphorous and sulfur. PVC contains chlorine. Nylon contains nitrogen. Teflon contains fluorine. Polyester and polycarbonates contain oxygen.
Polymers each have distinct traits but most polymers tend to have the same characteristics. Polymers tend to be resistant to chemicals, can be thermal and great electrical insulators, and for the most part are light-weight, with varying degrees of strength. They are also able to be made into a heavy walled jar, or a thin fiber.
Molecular Structure of Common Plastics